Academic Article, May 2026
Role of Hyperthermic Intraperitoneal Chemotherapy in Gynecologic Cancer Treatment
Asst.Prof. Marut Yanaranop, MD, PhD
Gynecologic Oncology Unit, Department of Obstetrics and Gynecology, Rajavithi Hospital
Cancers with peritoneal metastases including ovarian cancer, gastric cancer, and appendiceal cancer is generally associated with a poor prognosis and limited responsiveness to systemic chemotherapy (1). Hyperthermic intraperitoneal chemotherapy (HIPEC) combined with cytoreductive surgery (CRS), commonly referred to as “HIPEC surgery”, is a locoregional treatment to reduce disease recurrence and improve survival outcomes. However, HIPEC is a complex and high-risk procedure, necessitating careful and appropriate patient selection.
Pharmacologic principles of intraperitoneal (IP) chemotherapy
Peritoneal–plasma barrier limits the penetration of systemic chemotherapeutic agents into the peritoneal cavity (2). In contrast, IP chemotherapy achieves high local drug concentrations while reducing systemic toxicity. Drug penetration is approximately 1–3 mm. (3, 4). Hyperthermia at 41–43 °C is optimal for enhancing the cytotoxic effects of chemotherapy, which increases drug permeability, promotes greater intracellular accumulation in cancer cells, and disrupts cancer cell survival mechanisms (5, 6). Chemotherapeutic agents used in HIPEC are typically non–cell cycle-specific, can be maintained at higher concentrations within the peritoneal cavity than in plasma, and exhibit synergistic effects with heat. Common agents include cisplatin, carboplatin, oxaliplatin, mitomycin C, and doxorubicin (7, 8). HIPEC is performed in combination with CRS (9), aiming to eliminate both macroscopic tumors through surgical resection and residual microscopic disease through the heated chemotherapeutic agents within the peritoneal cavity.
Role of hyperthermic Intraperitoneal chemotherapy in gynecologic cancer treatment
HIPEC has emerged as an important therapeutic modality in the management of gynecologic malignancies. Its well-established role is in epithelial ovarian cancer (EOC), which frequently peritoneal metastasis and is associated with high recurrence rates despite standard treatment. For other gynecologic malignancies, such as malignant sex cord–stromal ovarian tumors, endometrial cancer, and cervical cancer, the available evidence remains limited (10)
Role of HIPEC in EOC treatment:
-
HIPEC in advanced EOC undergoing primary CRS: The role of HIPEC in this setting remains unclear. Although several retrospective studies have reported improved survival outcomes (11, 12, 13), the KOV-HIPEC-01 randomized controlled trial did not demonstrate a clear survival benefit associated with HIPEC (14). Therefore, HIPEC is not recommended as a standard treatment in this setting and should be considered only within the context of clinical trials
-
HIPEC in advanced EOC following neoadjuvant chemotherapy (NAC) and interval debulking surgery (IDS): This setting has the most robust supporting evidence. The OVHIPEC-1 trial demonstrated the efficacy of HIPEC in combination with IDS compared with IDS alone in patients with advanced EOC. The median progression-free survival was 14.2 months versus 10.7 months (HR: 0.66; 95% CI: 0.50–0.87; p = 0.03), and the median overall survival was 45.7 months versus 33.9 months, respectively (HR: 0.67; 95% CI: 0.48–0.95; p = 0.02), with no significant difference in severe adverse events (15). In addition, meta-analyses have supported the role of HIPEC in this context (16). Currently, HIPEC in this setting is supported for consideration in patients with advanced EOC who have received NAC and are candidates for successful IDS (17, 18). The recommended protocol includes 3–4 cycles of NAC followed by IDS and HIPEC using cisplatin at a dose of 100 mg/m², administered at 42–43 °C for 60–90 minutes, followed by adjuvant chemotherapy (19).
-
HIPEC combined with secondary CRS in recurrent EOC: The role of HIPEC in recurrent EOC remains controversial. Studies are heterogeneous and have yielded conflicting results (20, 21, 22). At present, HIPEC is not recommended as a standard treatment for recurrent EOC; however, it may be considered in carefully selected patients.
Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy procedure
1. Patient evaluation and selection: Patients suitable for HIPEC should have a good overall performance status, disease confined to the peritoneal cavity, and a high likelihood of achieving complete or near-complete cytoreduction.
2. Abdominal exploration and assessment of peritoneal dissemination: Systematic exploration of the abdominal cavity using diagnostic laparoscopy is performed to evaluate the peritoneal cancer index (23) to determine whether the patient is likely to benefit from surgical intervention. If adequate tumor debulking cannot be achieved, surgery may not confer a survival benefit and may instead increase the risk of unnecessary complications.
3. Cytoreductive surgery: CRS is performed with the goal of removing all visible tumor burden. It includes resection of involved organs (multivisceral resection), as well as hysterectomy and oophorectomy in patients with gynecologic malignancies. In addition, peritonectomy procedures are performed (9). This is a highly complex procedure, with an average operative time of approximately 8–12 hours. Surgical outcomes are assessed using the completeness of cytoreduction (CC) score (24), which is based on the size of residual tumor nodules, The CC score is directly associated with prognosis, overall survival, and risk of recurrence.
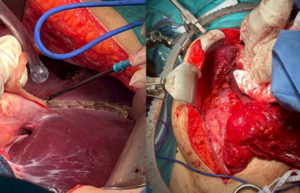 Figure 1 shows the surgical procedure of stripping the peritoneumbeneath the right diaphragm.
Figure 1 shows the surgical procedure of stripping the peritoneumbeneath the right diaphragm.
4. Hyperthermic intraperitoneal chemotherapy (HIPEC): Following completion of CRS, inflow and outflow catheters are inserted into the peritoneal cavity and connected to a perfusion pump that regulates both flow and temperature. Chemotherapeutic agents are heated to approximately 41–43°C and then circulated within the peritoneal cavity for 60–90 minutes. Then, the chemotherapeutic solution is drained from the peritoneal cavity, followed by peritoneal lavage. Surgical drains are placed, and the abdominal incision is closed.
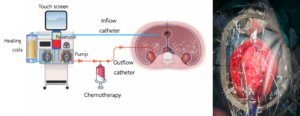
Figure 2 shows the connection for circulating the solution within the peritoneal cavity.
5. Post-operative care: The patient is transferred to an intensive care unit or a high-dependency unit for close monitoring.
Complications of hyperthermic intraperitoneal chemotherapy
HIPEC is associated with a reported complication rate of approximately 30–40%, while the mortality rate about 0-4% (25). Common complications include intra-abdominal or surgical site infections, anastomotic leakage, bowel obstruction or post-operative ileus, hemorrhage, thromboembolic events, as well as complications related to anesthesia and post-operative pain. In addition, adverse effects related to chemotherapeutic agents may occur, including nausea and vomiting, nephrotoxicity, and myelosuppression.
Conclusion
HIPEC is a locoregional treatment modality in patients with advanced EOC who have received NAC and can undergo successful IDS. Appropriate patient selection and treatment in specialized, experienced centers remain critical determinants of optimal outcomes.
Reference
-
Mohkam K, Passot G, Cotte E, Bakrin N, Gilly F-N, Ledochowski S, et al. Resectability of Peritoneal Carcinomatosis: Learnings from a Prospective Cohort of 533 Consecutive Patients Selected for Cytoreductive Surgery. Annals of Surgical Oncology. 2016;23(4):1261-70.
-
Dedrick RL, Myers CE, Bungay PM, DeVita VT, Jr. Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat Rep. 1978;62(1):1-11.
-
Jacquet P, Sugarbaker PH. Peritoneal-plasma barrier. Cancer Treat Res. 1996;82:53-63.
-
Tamura K, Kitayama J, Saga Y, Takei Y, Fujiwara H, Yamaguchi H, et al. Combined Intraperitoneal and Systemic Chemotherapy for Peritoneal Metastases: Drug Delivery Concepts, Pharmacokinetics, and Clinical Applications: A Narrative Review. Pharmaceutics. 2026;18(2).
-
Hildebrandt B, Wust P, Ahlers O, Dieing A, Sreenivasa G, Kerner T, et al. The cellular and molecular basis of hyperthermia. Crit Rev Oncol Hematol. 2002;43(1):33-56.
-
Löke DR, Helderman R, Franken NAP, Oei AL, Tanis PJ, Crezee J, et al. Simulating drug penetration during hyperthermic intraperitoneal chemotherapy. Drug Deliv. 2021;28(1):145-61.
-
Goodman MD, McPartland S, Detelich D, Saif MW. Chemotherapy for intraperitoneal use: a review of hyperthermic intraperitoneal chemotherapy and early post-operative intraperitoneal chemotherapy. J Gastrointest Oncol. 2016;7(1):45-57.
-
Valle SJ, Alzahrani NA, Liauw W, Sugarbaker PH, Bhatt A, Morris DL. Hyperthermic Intraperitoneal Chemotherapy (HIPEC) Methodology, Drugs and Bidirectional Chemotherapy. Indian J Surg Oncol. 2016;7(2):152-9.
-
Sugarbaker PH. Peritonectomy procedures. Ann Surg. 1995;221(1):29-42.
-
Margioula-Siarkou C, Almperis A, Papanikolaou A, Laganà AS, Mavromatidis G, Guyon F, et al. HIPEC for gynaecological malignancies: A last update (Review). Med Int (Lond). 2023;3(3):25.
-
Lei Z, Wang Y, Wang J, Wang K, Tian J, Zhao Y, et al. Evaluation of Cytoreductive Surgery With or Without Hyperthermic Intraperitoneal Chemotherapy for Stage III Epithelial Ovarian Cancer. JAMA Netw Open. 2020;3(8):e2013940.
-
Karanikas M, Kofina K, Kyziridis D, Trypsianis G, Kalakonas A, Tentes AA. HIPEC as Up-Front Treatment in Locally Advanced Ovarian Cancer. Cancers (Basel). 2024;16(20).
-
Ghirardi V, De Felice F, D’Indinosante M, Bernardini F, Giudice MT, Fagotti A, et al. Hyperthermic intraperitoneal chemotherapy (HIPEC) after primary debulking surgery in advanced epithelial ovarian cancer: Is BRCA mutational status making the difference? Cancer Treat Res Commun. 2022;31:100518.
-
Lim MC, Chang SJ, Park B, Yoo HJ, Yoo CW, Nam BH, et al. Survival After Hyperthermic Intraperitoneal Chemotherapy and Primary or Interval Cytoreductive Surgery in Ovarian Cancer: A Randomized Clinical Trial. JAMA Surg. 2022;157(5):374-83.
-
van Driel WJ, Koole SN, Sikorska K, Schagen van Leeuwen JH, Schreuder HWR, Hermans RHM, et al. Hyperthermic Intraperitoneal Chemotherapy in Ovarian Cancer. N Engl J Med. 2018;378(3):230-40.
-
Huo YR, Richards A, Liauw W, Morris DL. Hyperthermic intraperitoneal chemotherapy (HIPEC) and cytoreductive surgery (CRS) in ovarian cancer: A systematic review and meta-analysis. European Journal of Surgical Oncology (EJSO). 2015;41(12):1578-89.
-
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer (Version 2.2026). .
-
Filis P, Mauri D, Markozannes G, Tolia M, Filis N, Tsilidis K. Hyperthermic intraperitoneal chemotherapy (HIPEC) for the management of primary advanced and recurrent ovarian cancer: a systematic review and meta-analysis of randomized trials. ESMO Open. 2022;7(5):100586.
-
Aronson SL, Lopez-Yurda M, Koole SN, Schagen van Leeuwen JH, Schreuder HWR, Hermans RHM, et al. Cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy in patients with advanced ovarian cancer (OVHIPEC-1): final survival analysis of a randomised, controlled, phase 3 trial. Lancet Oncol. 2023;24(10):1109-18.
-
Ghirardi V, Trozzi R, Scanu FR, Giannarelli D, Santullo F, Costantini B, et al. Expanding the Use of HIPEC in Ovarian Cancer at Time of Interval Debulking Surgery to FIGO Stage IV and After 6 Cycles of Neoadjuvant Chemotherapy: A Prospective Analysis on Perioperative and Oncologic Outcomes. Ann Surg Oncol. 2024;31(5):3350-60.
-
Fagotti A, Costantini B, Fanfani F, Giannarelli D, De Iaco P, Chiantera V, et al. Hyperthermic Intraperitoneal Chemotherapy in Platinum-Sensitive Recurrent Ovarian Cancer: A Randomized Trial on Survival Evaluation (HORSE; MITO-18). J Clin Oncol. 2025;43(7):852-60.
-
Classe JM, Meeus P, Hudry D, Wernert R, Quenet F, Marchal F, et al. Hyperthermic intraperitoneal chemotherapy for recurrent ovarian cancer (CHIPOR): a randomised, open-label, phase 3 trial. Lancet Oncol. 2024;25(12):1551-62.
-
Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996;82:359-74.
-
Munoz-Zuluaga CA, King MC, Diaz-Sarmiento VS, Studeman K, Sittig M, MacDonald R, et al. Defining “Complete Cytoreduction” After Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy (CRS/HIPEC) for the Histopathologic Spectrum of Appendiceal Carcinomatosis. Ann Surg Oncol. 2020;27(13):5026-36.
-
Chen D, Ma Y, Li J, Wen L, Zhang G, Huang C, et al. Risk factors for postoperative complications in patients undergoing cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy: A meta-analysis and systematic review. Int J Colorectal Dis. 2024;39(1):167.







